 However, there remain several outstanding questions that suggest that this widely accepted interpretation requires closer examination and clarification. These data have led many in the field to believe that LRRK2, and the G2019S mutation in particular, inhibits growth. Previous work has shown that neurons grown in culture that overexpress LRRK2-G2019S have shorter neurites than non-transgenic neurons, while those cultured from mice with Lrrk2 deleted (KO) have longer neurites,. Several efforts have been made to identify predictable effects of altered LRRK2 levels or kinase activity in neurons grown in culture that could be used as bioassays as well as to pursue disease-related phenotypes in vivo. The G2019S mutation increases LRRK2 kinase activity, ,, , suggesting that therapies designed to reduce kinase activity may be relevant for treating PD however, the biological functions of LRRK2 are poorly understood. The structure of LRRK2 suggests that it is multifunctional it is a 286 kDa protein that contains several protein-protein interaction domains, a kinase domain, and a GTPase. The most common clinical mutation, G2019S, has been reported in almost 7% of familial PD patients with penetrance that is age-dependent–from 21% at age 50 to 81% at age 70. Mutations in the gene encoding leucine-rich repeat kinase 2 ( LRRK2) account for up to 13% of familial Parkinson's disease (PD) cases. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.Ĭompeting interests: The authors have declared that no competing interests exist. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.įunding: This work was supported by the Michael J. Received: DecemAccepted: MaPublished: April 30, 2013Ĭopyright: © 2013 Sepulveda et al. PLoS ONE 8(4):Įditor: Anna Dunaevsky, University of Nebraska Medical Center, United States of America Such predictable changes in motility can be exploited in LRRK2 bioassays and guide exploration of LRRK2 function in vivo.Ĭitation: Sepulveda B, Mesias R, Li X, Yue Z, Benson DL (2013) Short- and Long-Term Effects of LRRK2 on Axon and Dendrite Growth. Our findings support that LRRK2 can regulate patterns of axonal and dendritic growth, but they also show that effects vary depending on growth substrate and stage of development. 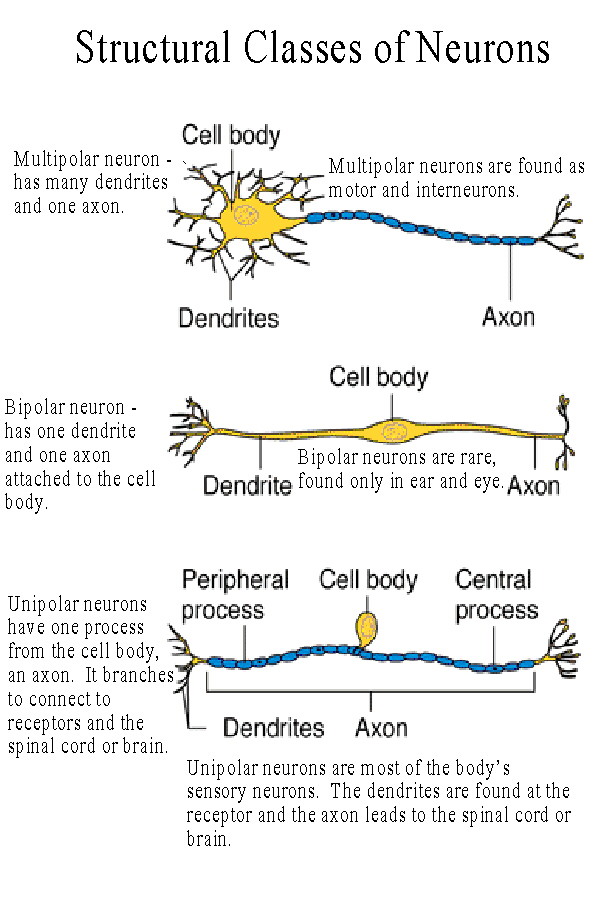
In contrast, young neurons cultured on a slower growth substrate, poly-L-lysine, show significantly reduced axonal and dendritic motility in Lrrk2 transgenic neurons and significantly increased motility in Lrrk2 knockout neurons with no significant changes in length. Over the course of three weeks of development on laminin, the data show a sustained, negative effect of LRRK2-G2019S on dendritic growth and arborization, but counter to expectation, dendrites from Lrrk2 knockout mice do not elaborate more rapidly. 
To address these questions, we compared several developmental milestones in neurons cultured from mice expressing bacterial artificial chromosome transgenes encoding mouse wildtype-LRRK2 or mutant LRRK2-G2019S, Lrrk2 knockout mice and non-transgenic mice. However, growth has not been measured it is not known whether mean differences in length correspond to altered rates of growth or retraction, whether axons or dendrites are impacted differentially or whether effects observed are transient or sustained. The function of LRRK2 is not well understood, but it has become widely accepted that LRRK2 levels or its kinase activity, which is increased by the most commonly observed mutation (G2019S), regulate neurite growth. Mutations in leucine-rich repeat kinase 2 ( LRRK2) underlie an autosomal-dominant form of Parkinson's disease (PD) that is clinically indistinguishable from idiopathic PD. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
